
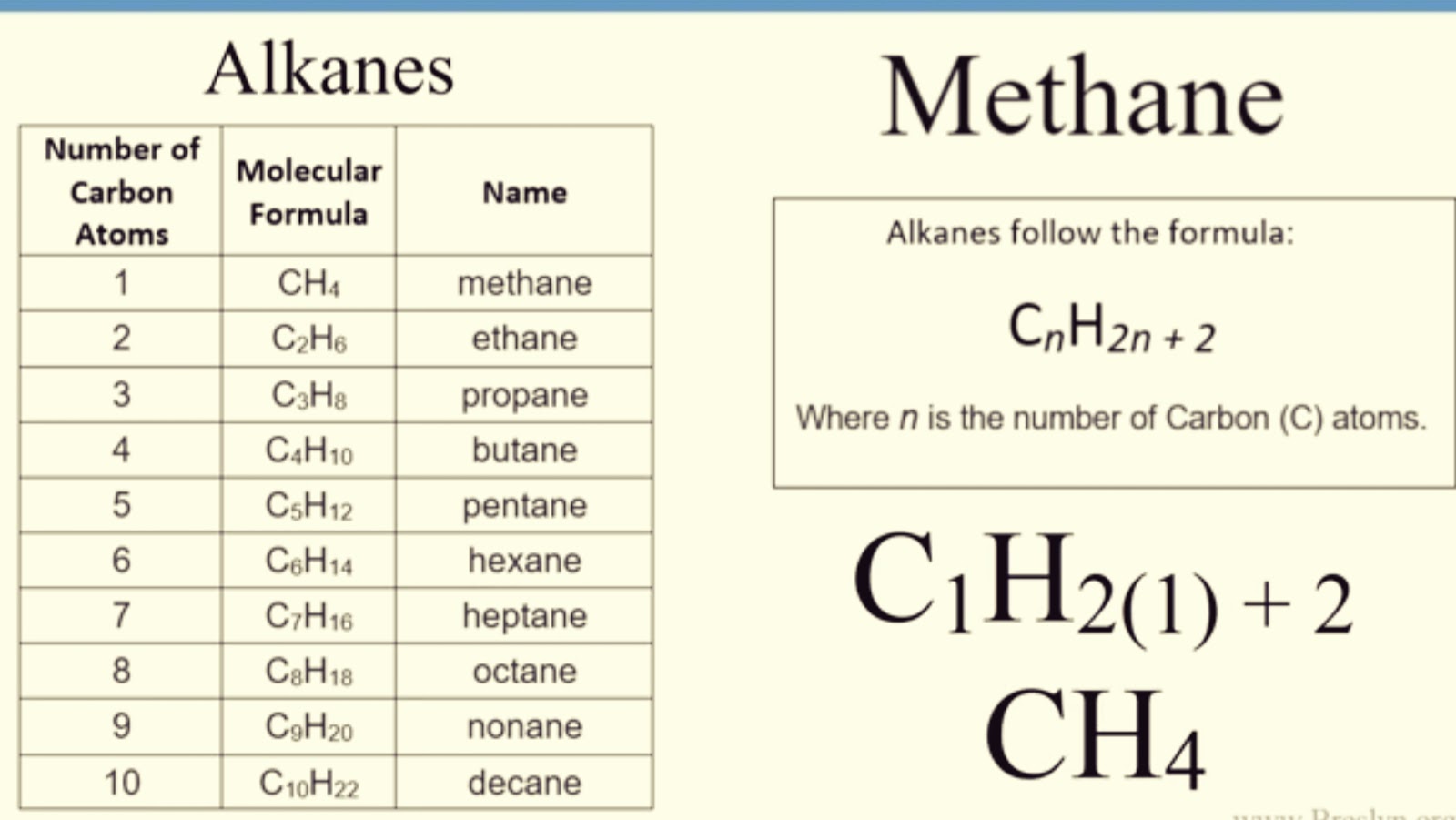
The products were identified and quantified by using GC–MS and GC-FID respectively. The fluxionality and structural diversity emerging at reaction conditions must be taken into account in theoretically descriptions of the catalytic interface.Ī simple and effective empirical method for carbonaceous substances production estimation and the effect of diluent N2 on it during pyrolysis of trifluoromethane (R23) was firstly studied at the temperatures of 973 K to 1223 K and residence times of 4 s to 40 s. The BO-layer transiently exposes the reactive -B=O motifs in the metastable states. Phase boundaries shift and blur, and phases consist of several stoichiometries and structures. The phase diagram of the interface constructed on the basis of the statistical ensembles of many accessible states is very different from the one based on global minima. The size of the thermal ensemble is composition-dependent. The interface has thermal access to multiple different stoichiometries, and multiple structures of each stoichiometry. We characterize the surface of the partially oxidized B-rich hBN surface under reaction conditions from first principles. Theoretically there is no limit to the length of hydrocarbon chains.Partially oxidized surfaces of hexagonal boron nitride (hBN) and several metal borides are unexpectedly excellent catalysts for oxidative dehydrogenation of alkanes to olefins, but the nature of the active site(s) on these B-containing interfaces remains elusive. The branched molecules shown here would be given names as follows. The removal of a hydrogen atom from a hydrocarbon molecule leaves an alkyl group that readily attaches to a functional group, or forms a branch on a hydrocarbon chain.įor example, CH 3 is named methyl CH 3 CH 2, ethyl CH 2 CH, ethenyl CH 3 CH 2 CH 2, propyl and so on.Ī benzene ring from which a hydrogen atom has been removed is often referred to as a phenyl. The double bonds become completely conjugated and no longer behave as double bonds. Heating ethane with steam is an important commercial process for making ethene (or ethylene). Hydrocarbons that contain one or more triple bonds are called alkynes, and is the name ending is yne. Like cycloalkanes, alkenes have the general formula C n H 2n. This breaks chemical bonds in the molecules, and forms smaller hydrocarbon molecules.Ĭracking is an example of a thermal decomposition thermal decomposition: A reaction in which substances are broken down by heat.

This chemical reaction produces smaller hydrocarbons, including alkanes and alkenes.Įthene and other alkenes are unsaturated unsaturated: An unsaturated compound contains at least one double or triple bond.Ĭracking Fuels made from oil mixtures containing large hydrocarbon hydrocarbons: Hydrocarbons are a group of compounds which contain the elements hydrogen and carbon. In organic chemistry, an alkene is an unsaturated hydrocarbon that contains at least one carboncarbon double bond.įractions that are produced by the distillation distillation: Distillation is when we make a liquid evaporate and then condense the vapour back to a purer liquid.
ALTERNATIVE BALANCED EQUATION FOR THE CRACKING OF DECANE CRACKED
One molecule of decane can be cracked to produce one molecule of ethene and. The heavier fractions can be split up into smaller molecules by catalytic cracking. The BBC is not responsible for the content of external sites.Ĭrude oil is first separated into fractions by fractional distillation. Get advice here Links Personalise your Bitesize Sign in, choose your GCSE subjects and see content thats tailored for you. They are then: heated to 600-700C passed over a catalyst of silica or alumina These processes break covalent bonds in the molecules, causing thermal decomposition reactions.Ĭracking of decane There is a greater demand for smaller hydrocarbons than larger ones.

